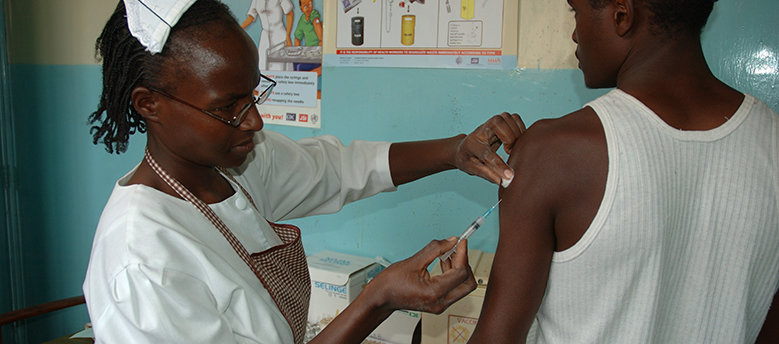
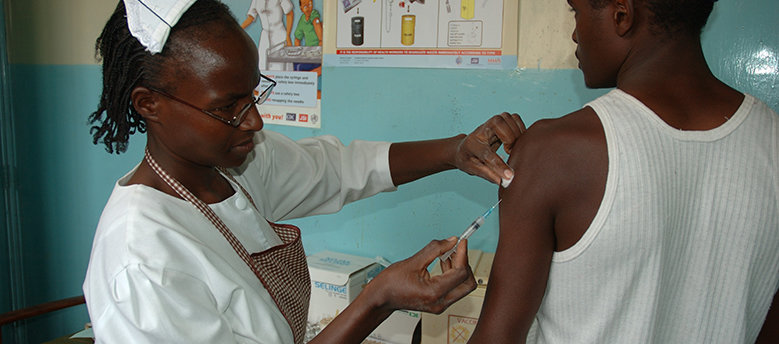
Possible breakthrough in tuberculosis research on the horizon
More than a century after the announcement of an alleged miracle cure against the disease, patients can now hope for new diagnostics and treatment approaches.
“New weapon in fight against the deadliest disease! Medicine can be used to treat existing illness and prevent reoccurrence.” These headlines were published 125 years ago and shook up not only the world of science and medicine, but also the general public. At the time, tuberculosis – the disease in question – was responsible for almost 40 percent of deaths among Berlin’s working population.
Author: Stefan H. E. Kaufmann
Robert Koch, who discovered the miracle cure, had already described the infectious nature of tuberculosis nine years earlier – on 24 March 1882 to be precise, which is why 24 March was declared World Tuberculosis Day. But that was nothing compared to his new miracle drug tuberculin. If the first reports about the successful treatment of consumptives are to be believed, it was an unparalleled success story. The results of the clinical studies on the chances of recovery for tuberculosis patients were published at the end of the year. The euphoria abruptly turned into disappointment, however, as the studies showed that the treatment was not effective.
Up to a short time ago, very little had changed since then. Of course, 95 years ago, French researchers Albert Calmette und Camille Guérin developed a tuberculosis vaccine, the Bacille Calmette-Guérin (BCG) vaccine which is named after them, and 20 years later, Salman Waksman discovered streptomycin, the first effective drug for the treatment of tuberculosis. However, the vaccine only protects young children against serious disease progression and many tuberculosis pathogens are already resistant to streptomycin.
Do we still have to concern ourselves with this today? In my opinion we do: tuberculosis has claimed in excess of one billion human lives over the last 200 years, more than all other communicable diseases, and more than smallpox, malaria, the plague, influenza, cholera and AIDS combined over the same period. Even today, no pathogen is responsible for more deaths than the forgotten tuberculosis pathogen, Mycobacterium tuberculosis. Yet despite these dreadful numbers, tuberculosis is a forgotten disease that lacks the shock value and media hype of more recent diseases like Ebola and Zika.
This is aggravated by the fact that tuberculosis bacteria are increasingly resistant to the antibiotics used to treat the disease. The treatment of normal tuberculosis is extremely complicated and protracted. Up to four drugs have to be taken for six months. In the places where tuberculosis is most widespread, i.e. in the world’s poorer countries, there is no guarantee of orderly treatment. And this makes it even easier for pathogens that are resistant to several drugs at once to develop.
Against this background, it could be said that the World Health Organization’s aim of reducing the annual disease rate from 9.6 million cases today to fewer than one million, and the number of fatalities from 1.5 million today to 75,000 in the year 2035 is far too ambitious. The target is ambitious - but it is not entirely unrealistic.
Research and development in the field of tuberculosis was neglected for almost 100 years. It only took off again towards the end of the 20th century and we are now able to harvest the first fruits of these efforts today. In 2014, two new drugs were licensed for the treatment of tuberculosis– not for general use but for the treatment of multiresistant strains, the cases for which drugs are most urgently needed. Five other drugs and several new combinations of drugs are currently close to the end of clinical testing. Thus, the tuberculosis drug pipeline is much fuller now than it has been for many years.
Progress has also been made in the area of diagnostics. Using the GeneXpert system, not only can infection with the pathogen be diagnosed very quickly with the help of molecular genetic methods, it is also possible to analyze its resistances. Patients can therefore now be put on suitable treatment in a matter of a few hours. GeneXpert yields the best results when pathogenic material can be harvested from the patient. This is usually possible in the case of the most common form of the disease, pulmonary tuberculosis. However, it does not work in cases in which the lungs are not affected – around 20 percent of all patients.
The tuberculin test originally developed by Robert Koch does not help here either, as it does not differentiate between those who have the disease and healthy infected subjects. There are over two million people in the world infected with the pathogen but who do not suffer from the disease. Active tuberculosis only arises in just under ten percent of all those infected.
Biomarker tests, for which blood cells are used, offer a solution here. Doctors will soon be able to not only diagnose the disease using such biomarkers but also provide a prognosis. In other words, not only can we distinguish sufferers from healthy infected subjects, but also whether an infected person is at a high risk of developing active tuberculosis or not. Similar to the way in which biomarkers can be used in the diagnosis of different forms of cancer, people with an increased risk of developing tuberculosis can now also be identified. In people identified as being at risk in this way, the development of the disease can be prevented through the administration of preventive drug treatment. Because they cannot become infectious as a result, this also reduces the risk of the spread of the disease.
With over a dozen candidates at different stages of clinical testing, the pipeline for tuberculosis vaccine research and development is also flowing. The immunization effect of two candidates is currently being tested, and a new vaccine is due to enter the final stage of clinical testing in India this year. As part of this study, it is intended to examine whether it is possible to prevent the recurrence of the disease following successful drug treatment. Although the treatment of tuberculosis with drugs generally leads to recovery, every tenth person discharged as recovered suffers a recurrence of the disease. The new study should establish whether the vaccine can prevent this. If it works, it would represent a decisive breakthrough!
These new developments are gratifying and, for the first time in many years, give us hope that we will succeed in getting tuberculosis under control in the early 21st century.