Chromatin and immunity
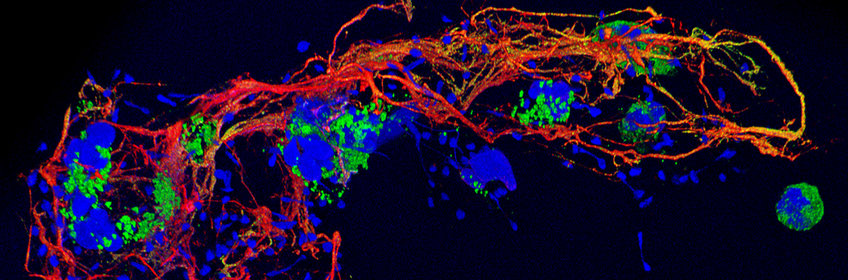
Neutrophils and NETs
Most of the white blood cells of humans are neutrophils. They have different strategies for fighting microbes, and we are especially interested in one of them: Neutrophil Extracellular Traps (NETs). NETs are extracellular structures made of chromatin and specific proteins. They are released from the neutrophil after unique cell death programs and are essential for trapping and killing even large microorganisms such as fungi. NETs also initiate coagulation and can activate cells of the immune system. We are interested in understanding the mechanisms of NET formation as well as the function of NET components, especially histones.
Histones in immunity and development
Histones have a double function in immunity: they regulate gene expression and play a direct role as antimicrobial components. We study both aspects of histone function. In order to understand how histones contribute to transcriptional and epigenetic regulation we use the model system Drosophila melanogaster. This system provides the unique opportunity to elucidate structure-function relationships for histones. We also systematically address the structural and physiological requirements for the antimicrobial activity of histones in the context of chromatin and NETs. We are testing the hypothesis that the two seemingly unrelated functions of histones are evolutionarily interlinked.